Jim’s Big-Ass Retirement Party 2025
By Rebecca Smith

Welcome sign for the big day!
When Jim Murphy, Chairman and President Emeritus, decided to hang up his hat at the end of the year, we knew there was going to be a bigtime celebration in our near future. And come February, we didn’t need to twist any arms to have a healthy turnout of well-wishers to send him off into retirement!

David and Christopher, front and center during the staff videos and slideshow.
Each of Pickering’s employees deeply wanted to spend time sharing a memorable story and cheers with our dear Mr. Murphy, so in addition to our surprise farewell video, Jim was very busy during the party chatting with all of our guests. When I spoke with him after it wrapped, he was overwhelmed with joy and gratitude for these relationships, cultivated over the decades.

Jim with Gloria, our longest tenured employee, who has known Jim for 33 years and counting!

Jim with Severo, Anita and her husband Paul. All friends and colleagues for over two decades.
If you’ve had the opportunity to meet Jim during his 33 years at Pickering, whether you met him at Pittcon in the earlier years or have been by the lab, you’ve likely come to respect and admire him. He is compassionate and has a great sense of humor. Jim asks about your family, remembers details in your life, and he’ll help you put air in your tires (ask me how I know). Jim honored our company’s rich history, but he’s committed to Pickering’s future, and as such, steadily built his succession plan for over a decade.
During his tenure, Jim evolved Pickering Labs into the thriving company it is today from the groundwork of founder Dr. Michael Pickering’s vision. Jim started in 1991, about a decade into Michael’s greatest experiment (as he called the lab). And as the early partnership formed, Michael created more new products and met with customers, while Jim secured the people, resources, and finances to execute and support the business’s growth. Ten years in, this fearsome duo worked together to bring Pickering Labs into it’s forever home at 1280 Space Park, and within our walls, we further blossomed.

Throwback to Jim and the team celebrating our first Onyx PCX ready to be shipped to the customer!

Jim and Michael in the earlier years.
What a blast! Judy Pickering was not going to miss Jim’s big party!
Throughout the years of Jim’s tenure, the Pickering team has relied on him. He has been steadfast and true, respectful and welcoming. He’s shepherded many an instrument through the expensive design and launch phase, tripled the revenue of the company, not to mention interviewed and hired most of our employees, past and present! Jim has put up with our (my) quirks, he’s challenged us for our best work, and he has touched all of our hearts.

Jim with his wife Michelle, and Rebecca with my husband, Paul. I’ll dearly miss my daily chats with my longtime mentor.
Please allow us this chance to celebrate with you and share our love for this man who has so powerfully impacted our lives. Jim’s ensured Pickering’s future with his long-planned and thoughtful transition, and we in turn will do our part in forging ahead to our future.
Jim, we will always be your family and you’ll forever have a second home at 1280 Space Park! We love you and profoundly appreciate everything we have learned from you. You are cherished. We miss you already… and we cannot wait to see you onsite next month for the Board meeting. 😉

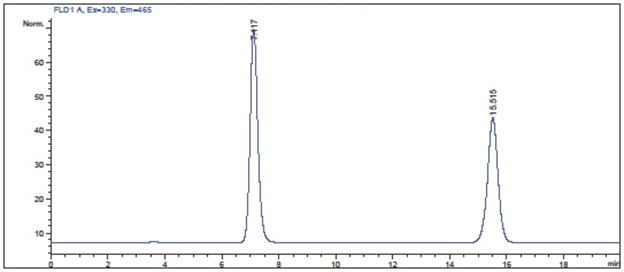
 observing some discoloration in the piston wash solution. Under normal circumstances, the solution has no color and now the solution is yellowish and the piston wash volume continues to increase.
observing some discoloration in the piston wash solution. Under normal circumstances, the solution has no color and now the solution is yellowish and the piston wash volume continues to increase. Our winners will shortly be receiving: a $100 gift card from Amazon.com!
Our winners will shortly be receiving: a $100 gift card from Amazon.com!