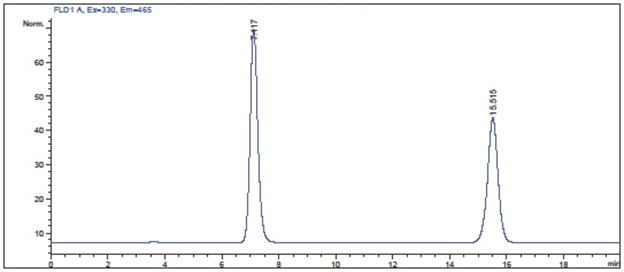
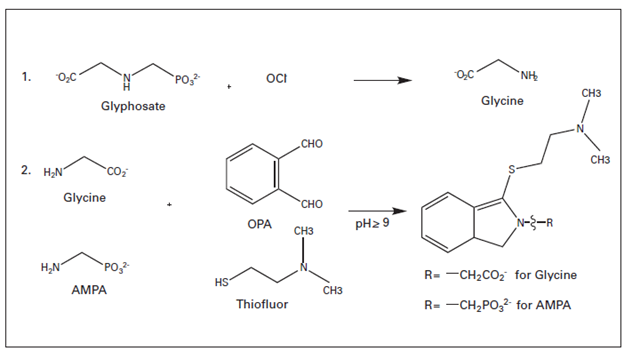
Pickering Laboratories – Chromatography Quiz
Announcing the winners of our previous quiz, Chromatography Quiz #47 – Piston Washed Out
Pickering Laboratories would like to congratulate the winners of our previous newsletter’s Chromatography Quiz #47: Marissol Mallon from Suffolk County Water Authority, Lakeisha Packer from Pennsylvania Department of Agriculture, Tom Schneider from Suffolk County Water Authority, Josiah Hakala from Minnesota Department of Health, and Narjes Ghafoori from Los Angeles County Public Health Laboratory.
Our winners will shortly be receiving a Reclaimed Wood Herb Garden, just in time for Spring! An herb garden with three fragrant pre-planted herbs: rosemary, thyme, and sage. Planted in a rustic reclaimed wood crate, to nestle perfectly on a sunny windowsill or kitchen counter. Happy growing!

Congratulations to our quiz winners! Thank you all for your submissions!
Chromatography Quiz #47 Solution: Discoloration in the piston wash is usually an indication that reagent has leaked into the piston wash bottle. The backend of the piston of the pump is directly connected to the piston wash. If the main piston seal is bad, you can have a small amount of reagent leak into the piston wash bottle.
Discoloration in the piston wash is usually an indication that reagent has leaked into the piston wash bottle. The backend of the piston of the pump is directly connected to the piston wash. If the main piston seal is bad, you can have a small amount of reagent leak into the piston wash bottle.
It is recommended to change the pump seals and then perform a pressure test to make sure the seal integrity is good.
Quiz #48 – Valve Lost Error

Here is a question for our Pinnacle and Onyx PCX users. A possible error for your system is a “Vale Lost” error. The valve assembly uses optical sensors to determine which position the valve is in. If the valve is unable to arrive at a programmed position, you will receive a Valve Lost Error.
What can lead to a Valve Lost Error?
Submit your answers to rsmith@pickeringlabs.com by April 17, 2026!