By Maria Ofitserova
 As novel Covid-19 virus spreads around the globe, pharmaceutical companies worldwide are racing to develop vaccines, therapeutic agents and prophylactics to combat the disease. A lot of these pharmaceuticals are biologics – in other words, they are either isolated from natural sources or manufactured via biotechnology methods.
As novel Covid-19 virus spreads around the globe, pharmaceutical companies worldwide are racing to develop vaccines, therapeutic agents and prophylactics to combat the disease. A lot of these pharmaceuticals are biologics – in other words, they are either isolated from natural sources or manufactured via biotechnology methods.
The focus on biological products during the current pandemic has highlighted the crucial role Amino Acids Analysis plays in the research and production of pharmaceuticals. As a result, Pickering Laboratories is seeing a surge in demand from customers requiring the consumables necessary to support these critical efforts.
Amino Acids Analysis can be used for:
- Identification tests for peptides and proteins by means of amino acids composition analysis;
- Determination of Impurities and related substances in Active Pharmaceutical Ingredients (APIs) and intermediates;
- Single or total amino acids quantification in drug products, including markers determination in complex matrixes;
- Control of manufacturing processes for recombinant proteins.
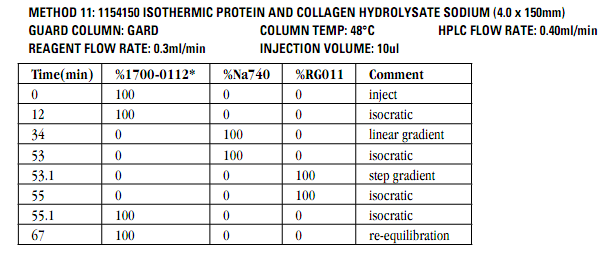
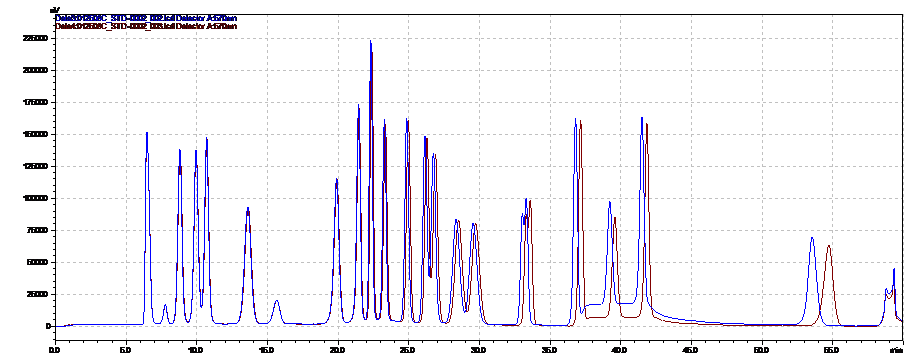
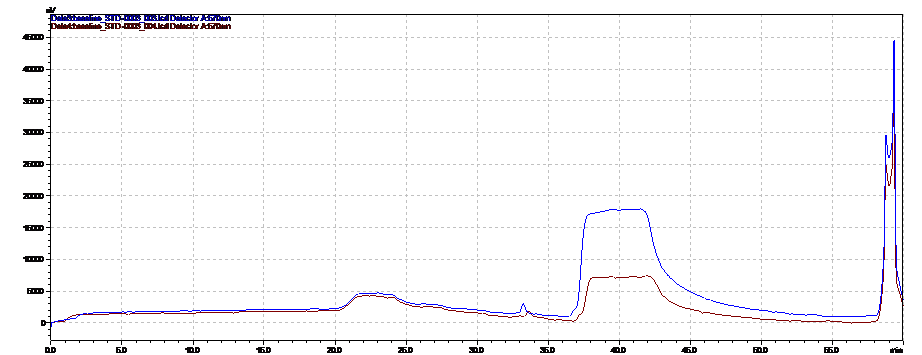
Our Amino Acids Analysis methods allow laboratories to perform testing according to European Pharmacopeia as well as testing of hydrolyzed and native substances.
Though it seems the focus is now set firmly on pharmaceutical industry, there are other vital areas of amino acids testing that cannot be overlooked. No less important is the Amino Acids Analysis of physiological samples done by hospitals and clinical laboratories. Testing of newborns for inborn errors of metabolism, such as PKU and MSUD, as well as monitoring amino acids markers for different medical conditions, including rejection of organ transplants, metabolic disorders and diabetes, are all tests that still have to be done in a timely manner to prevent adverse health outcomes, pandemic or not.
Preventing disruption in food supply is another area that needs amino acids analysis to continue uninterrupted during the global Coronavirus crisis. Feed and food testing laboratories are already under stress due to special quarantine requirements, so providing a steady supply of reagents and chemicals is another way Pickering doing its part in keeping these industries running.
As an expert in Amino Acids Analysis and a trusted supplier of related consumable products, Pickering Laboratories is focused on supporting our new and existing customers as they navigate increased demand for testing. We are open for business and operating at full production capacity during the pandemic.
During these trying times, we are proud to continue providing timely product delivery and technical assistance to help ensure uninterrupted operations for essential children’s hospital, clinical laboratories, pharmaceutical companies and their testing laboratory partners, not to mention drinking water and food testing labs worldwide with our other post-column applications.
To learn more or ask specific questions about our product availability and up-to-date lead times, please contact us at orders@pickeringlabs.com or support@pickeringlabs.com and we will gladly address your concerns.
 If you are interested, feel free to check out some cool work that Thermo Scientific published on behalf of RSSL in the United Kingdom, using an ICS-6000 in combination with a Pickering post-column system to perform Amino Acids Analysis for the Pharmaceutical Industry.
If you are interested, feel free to check out some cool work that Thermo Scientific published on behalf of RSSL in the United Kingdom, using an ICS-6000 in combination with a Pickering post-column system to perform Amino Acids Analysis for the Pharmaceutical Industry.
 No other analytical technique, including pre-column derivatization followed by reversed-phase chromatography, has been shown to match post-column ion-exchange methods in accuracy and reproducibility. This is because the retention mechanism in ion exchange provides for chromatography that is almost completely matrix-insensitive. Simple sample preparation for native samples is an added benefit of the ion-exchange method.
No other analytical technique, including pre-column derivatization followed by reversed-phase chromatography, has been shown to match post-column ion-exchange methods in accuracy and reproducibility. This is because the retention mechanism in ion exchange provides for chromatography that is almost completely matrix-insensitive. Simple sample preparation for native samples is an added benefit of the ion-exchange method. As novel Covid-19 virus spreads around the globe, pharmaceutical companies worldwide are racing to develop vaccines, therapeutic agents and prophylactics to combat the disease. A lot of these pharmaceuticals are biologics – in other words, they are either isolated from natural sources or manufactured via biotechnology methods.
As novel Covid-19 virus spreads around the globe, pharmaceutical companies worldwide are racing to develop vaccines, therapeutic agents and prophylactics to combat the disease. A lot of these pharmaceuticals are biologics – in other words, they are either isolated from natural sources or manufactured via biotechnology methods.
 Biopharmaceuticals are large molecules produced by or extracted from biological sources. They are used for therapeutic as well as diagnostic purposes and include recombinant proteins, antibodies, vaccines, blood factors, hormones and many other types of substances. Currently, there are more than 200 biologics on the market and they account for almost a third of all pharmaceuticals under development.
Biopharmaceuticals are large molecules produced by or extracted from biological sources. They are used for therapeutic as well as diagnostic purposes and include recombinant proteins, antibodies, vaccines, blood factors, hormones and many other types of substances. Currently, there are more than 200 biologics on the market and they account for almost a third of all pharmaceuticals under development.
 Laboratories, Narjes Ghafoori from LA County Environmental Toxicology Lab, Joy Gottlieb from New Mexico Department of Health Scientific Lab Division, Hossein Hajipour from Texas Dept. of State Health Services Laboratories, and Widchuda Meeim from Thailand Bureau of Quality Control of Livestock Products.
Laboratories, Narjes Ghafoori from LA County Environmental Toxicology Lab, Joy Gottlieb from New Mexico Department of Health Scientific Lab Division, Hossein Hajipour from Texas Dept. of State Health Services Laboratories, and Widchuda Meeim from Thailand Bureau of Quality Control of Livestock Products.