Chromatography Quiz #39 Rising Baseline – Announcing our Winners!
Pickering Labs would like to congratulate all of our winners for our previous newsletter’s Chromatography Quiz:

Uwe Aulwurm from LCTech GmbH, David Green from Pepperdine University, Narjes Ghafoori from Los Angeles County Public Health Laboratory, Josiah Hakala from Minnesota Department of Health, and Tom Schneider from Suffolk County Water Authority.
Winners will soon receive a Springtime Bakery Tray! Wooden tray filled to the rim with an assortment of delightful baked goods!
Congratulations to our quiz winners! Thank you all for your submissions!
Answer to Previous Quiz:
What is causing the Carbamates baseline to rise?
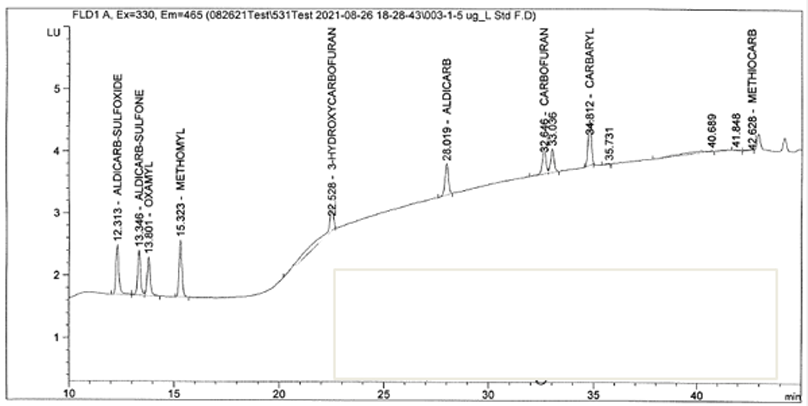
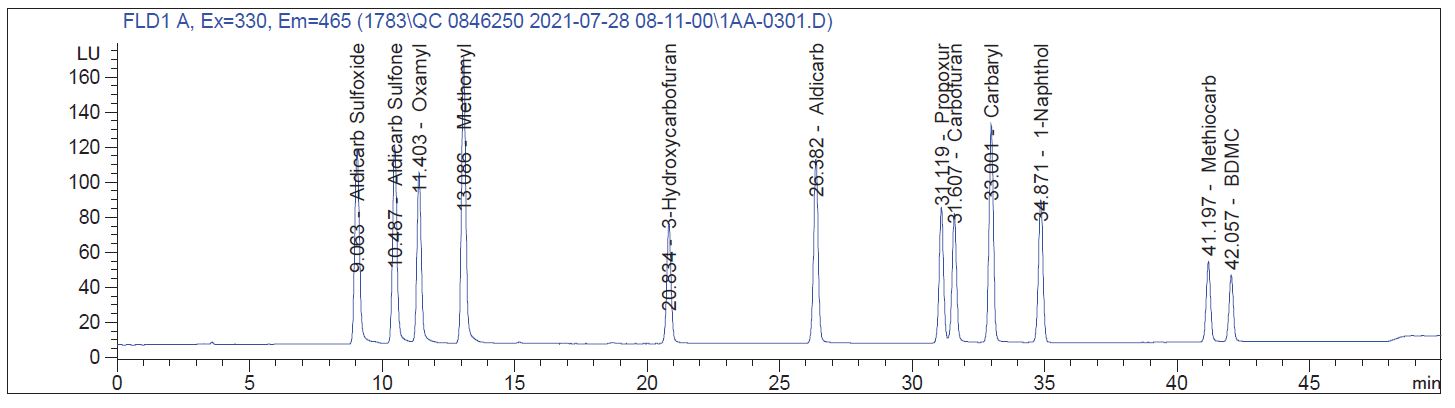
Our customers have been encountering this problem a lot recently. The problem is coming from the methanol (channel B). As the percentage of methanol increases, the baseline increases as well. This problem seems to be compounded due to supply chain issues. We recommend getting a new lot of methanol from your vendor and obtaining LCMS grade. If the new lot does not help, try a new vendor. You will most likely need to flush out your HPLC pump to fully resolve this issue. As always, please refrain from using filters in your mobile phase bottles, as this can take contamination from one bottle to the next. Contact support@pickeringlabs.com with any further questions.
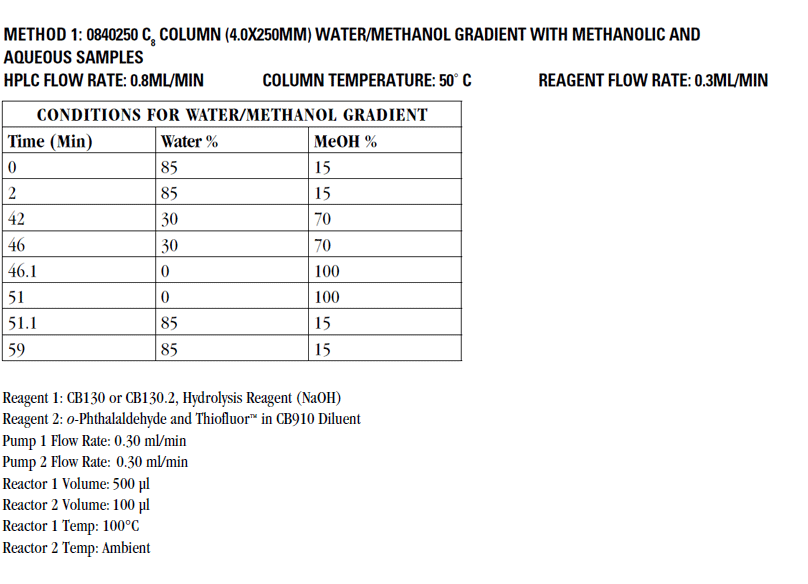
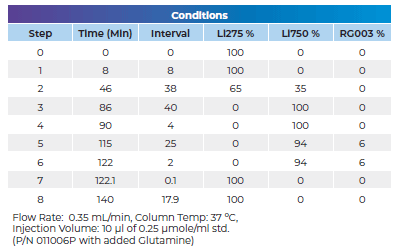
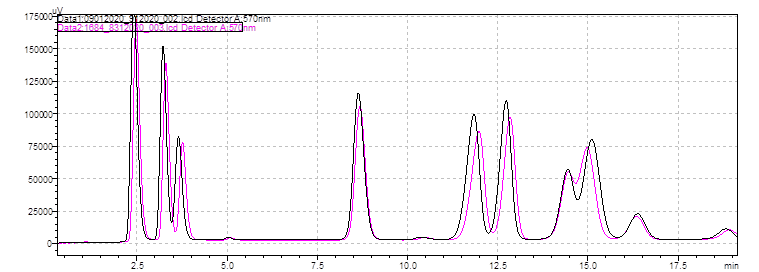
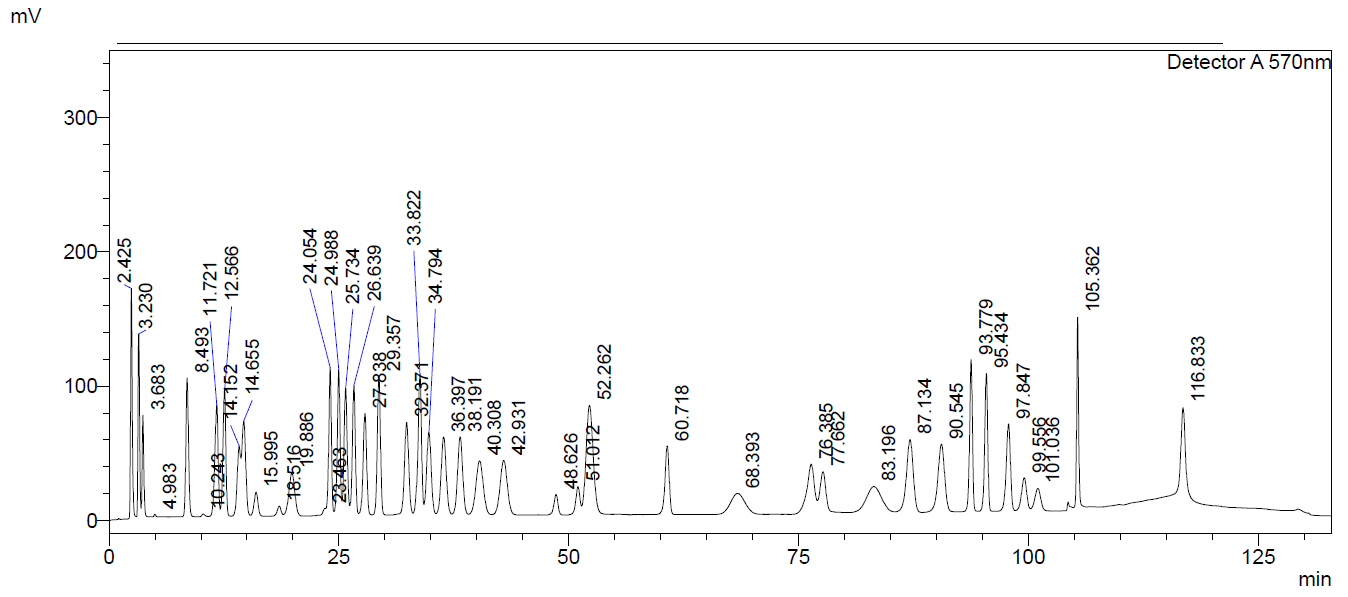
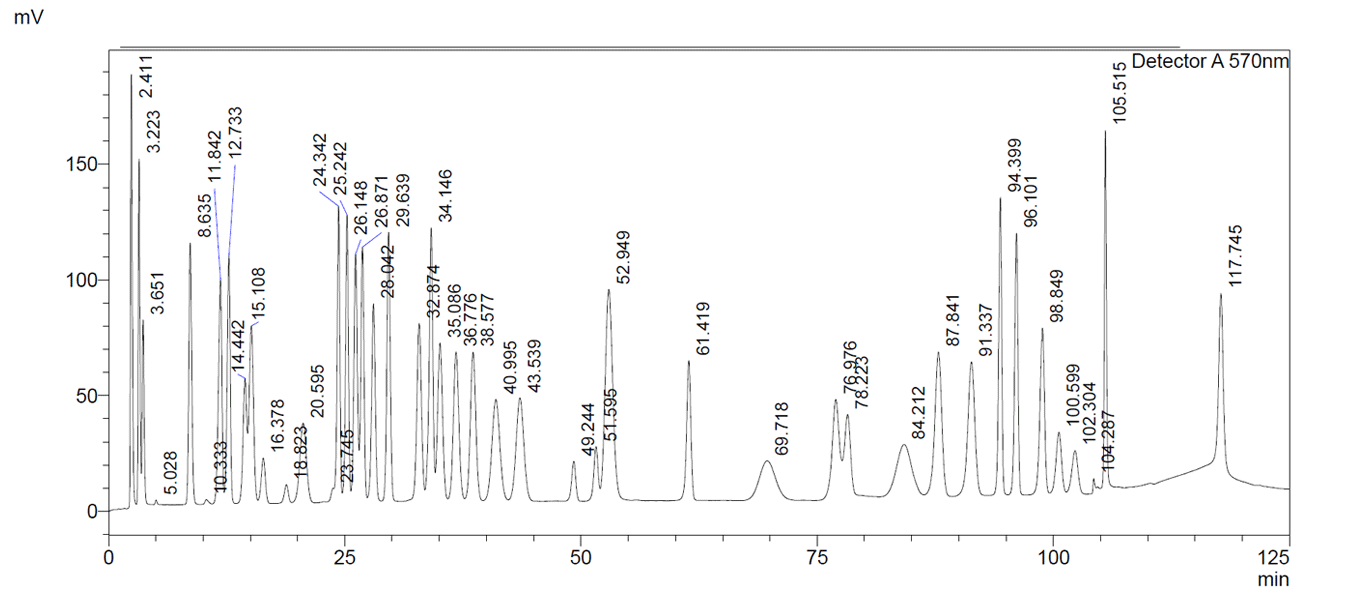
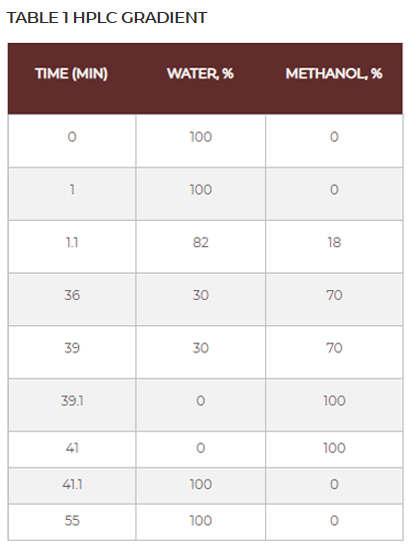
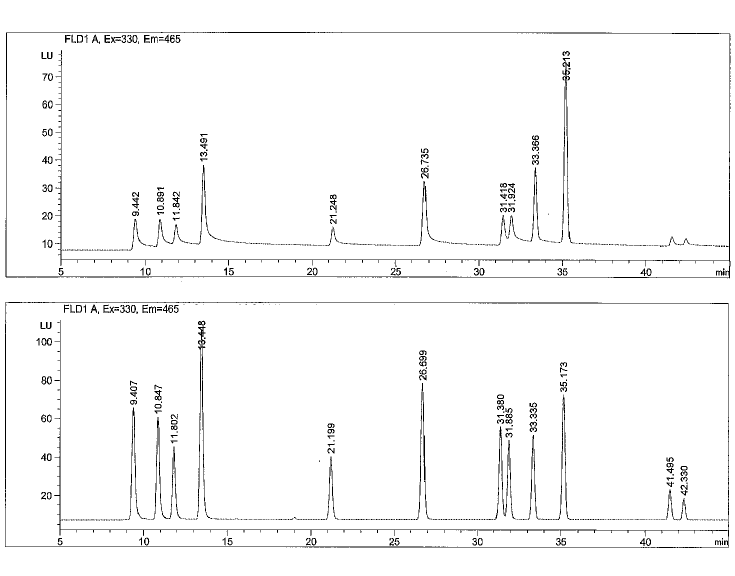
Below you will find a reference chromatogram, HPLC gradient table, and bad chromatogram as a refresher from the last quiz. Keep scrolling to find this quarter’s fun new quiz!
Bad Chromatogram
Good Chromatogram
If you have difficulty reading the images for Quiz #39, please click on them to enlarge or contact David Mazawa (david@pickeringlabs.com) to obtain a more user friendly PDF.
Chromatography Quiz #40 – Trivia for Pickering’s 40 Year Anniversary (multiple choice)
Check out our 40th Anniversary Timeline and complete the quiz below to win a prize. Simply email your answers and your full contact information to Rebecca at rsmith@pickeringlabs.com by April 29, 2022 in order to win. You will receive email confirmation when your submission is received, and winner congratulations will be published in the next issue (to be anonymous, please notify Rebecca in submission).
1. When did Pickering Labs move to Space Park Way?
a. 1998
b. 1999
c. 2000
d. 2001
2. Michael Pickering was granted a United States trademark for the name _________.
a. Thiofluor
b. Mycotox
c. Trione
d. Restore
3. Which of the following was the first “product testing solution” produced by Pickering Labs?
a. Saliva
b. Lung Fluid
c. Urine
d. Perspiration
4. Pickering Laboratories won the AOAC Single Laboratory Validation Method of the Year for the following method:
a. Amino Acids
b. Ionophores
c. Cannabinoids
d. Mycotoxins
5. Michael Pickering was issued a United States Patent for the following product:
a. Thiofluor reagent
b. Ninhydrin reagent
c. Restore solution
d. Vanillin reagent
6. Which post-column system was introduced in the year 2006 as the lower cost option similar to the PCX 5200?
a. Vector PCX
b. Pinnacle PCX
c. CRX400
d. Onyx PCX
7. Which year did Pickering move to their current location at 1280 Space Park Way?
a. 1997
b. 1998
c. 1999
d. 2000
8. Which year was the Onyx PCX introduced?
a. 2018
b. 2019
c. 2020
d. 2021
9. Our Eccrine Perspiration solution was first developed by the request from ______.
a. Crime and Punishment
b. Crime Sciences Inc.
c. Crime Scene Investigation
d. True Detective
10. Pickering has been supporting the scientific community for nearly 30 years by continuously exhibiting at the following Conference:
a. ACS
b. AOAC International
c. Florida Pesticide Residue Workshop
d. Pittcon
__________________________________________
David Mazawa
Technical Support Chemist
Pickering Laboratories, Inc.
1280 Space Park Way
Mountain View, CA 94043, USA
david@pickeringlabs.com
Phone: (650) 694-6700 x710



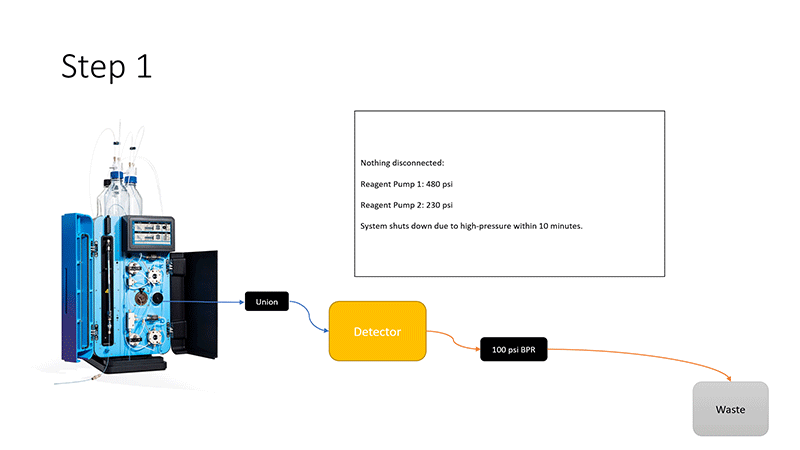

 Pickering Labs would like to congratulate all of our winners for our previous newsletter’s I’m not seeing any peaks! Carbamates edition Quiz: Tom Schneider from Suffolk County Water Authority, Narjes Ghafoori from LA County Environmental Toxicology Lab, and Jiufeng Fan from Glaxo Smith Kline, and Dr. David Green from Pepperdine University.
Pickering Labs would like to congratulate all of our winners for our previous newsletter’s I’m not seeing any peaks! Carbamates edition Quiz: Tom Schneider from Suffolk County Water Authority, Narjes Ghafoori from LA County Environmental Toxicology Lab, and Jiufeng Fan from Glaxo Smith Kline, and Dr. David Green from Pepperdine University.
